Partner for the administration of your life science companies.
Are you an investor? Different stages of development have different requirements that need the full attention of the team. In addition, you expect your fund’s reporting requirements and deadlines to be met.
For the important phases in your startup, we have service packages that we tailor exactly to your needs:


Preparation & Financing
- Business plan check
- Shareholder, investment contracts
- Establishment of bank relationship
- Coordination of investors, lawyers
Foundation
- Preparation of founding documents
- Articles of association, partnership agreement
- Shareholder contracts, investment contracts
- Coordination (of documents) with external partners: tax consulting firm, law firm
- Setting up systems and SOPs
- Budgeting and controlling processes
- Insurance


Ongoing Administration & Finance
- Accounting, preparation of annual financial statements
- Ongoing payroll accounting
- Controlling, cost centers, reporting
- Shareholder meetings, supervisory board
- Notaries
- Insurances
Standards jungle? Quality Management ‘as a Service’
- Analysis and creation of technical documentation
- Coordination of product testing
- Certification of your QM system
- Regulatory consulting
For example, in 2020, Fetter + Spiritini Group launched its first medical face mask with Activoris support (www.fesanocare.de).

WE ARE LOOKING FORWARD TO YOUR CHALLENGE!
E.Mail: info@activoris.com TEL: +49 (0)6453.585 35 00
Regulatory Affairs: Understanding requirements holistically – the “CE” for your products

- Specifications, risk analysis
- Coordination of test institutes
- Product validations, usability studies
- Clinical evaluation
- Technical documentation
Our passion: Drug Device Combinations
The top tier of regulatory affairs. Regulatory approval of combination products requires knowledge and experience in medtech and drug regulatory pathways. Our decades of experience with delivery systems for inhaled drugs and injection systems is thus a valuable wealth of knowledge for your drug-device combination.

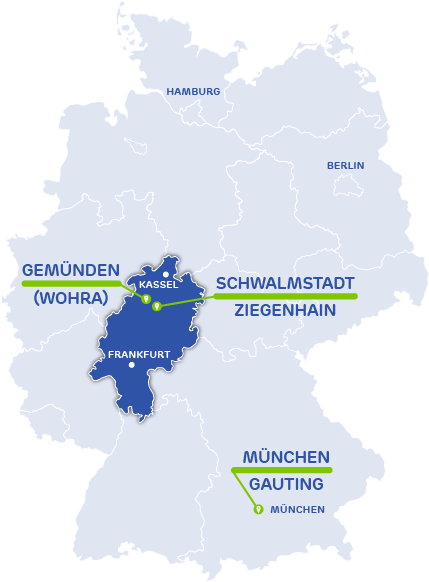
Activoris Medizintechnik GmbH
Seat, Adinistration, Devlopment, Production
Wohraer Str. 37
35285 Gemünden (Wohra)
Germany
+49.6453.58535.0
+49.6453.58535.25
Development Team Munich
Robert-Koch-Allee 29
82131 Gauting
Germany
+49.6453.58535.61
+49.6453.58535.25
Activoris Food Packaging GmbH
Seat & Administration
Wohraer Str. 37
35285 Gemünden (Wohra)
Germany
+49.6453.58535.0
+49.6453.58535.25
Production
An der Lämmerweide 10
34613 Schwalmstadt
+49.6691.91230.0
+49.6691.91230.25



